|
Researchers used computer simulations to screen
millions of molecules for opioid-like pain-relieving
properties to create a molecule (PZM21) that
effectively alleviates pain, but with fewer side
effects than the opioid morphine, in mice.
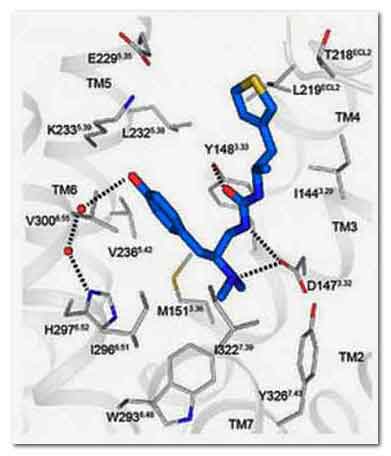
A computer model of the synthesized pain relieving
compound PZM21 (blue) docked with the mu opioid
receptor (grey).Dr. Bryan Roth, University of North
Carolina
Opioids are a class of powerful pain-relieving drugs
that work by activating opioid receptors on nerve
cells in the body and brain. These pain relievers
are generally safe when taken for a short time and
as prescribed by a doctor, but are frequently
misused because they also produce euphoria. When
misused or abused, opioid pain relievers can be
addictive and dangerous.
Scientists have identified 3 types of opioid
receptors: mu, delta, and kappa.
The mu receptors are responsible for opioids’
pleasurable effects and ability to relieve pain.
Studies suggest that once activated, the mu receptor
turns on 2 signaling pathways. One pathway, mediated
by the G protein Gi, underlies opioids’
pain-relieving properties. The other, mediated by
the beta-arrestin protein, leads to the undesirable
side effects of opioids, such as constipation and
slowed breathing.
To search for a potential pain reliever with fewer
side effects than current opioids, a research team
led by Dr. Bryan Roth at the University of North
Carolina and Dr. Brian Shoichet at the University of
California, San Francisco, screened more than 3
million compounds for those that may be able to turn
on the Gi-mediated pathway, but not beta-arrestin.
The study was funded by NIH’s National Institute of
General Medical Sciences (NIGMS), National Institute
on Drug Abuse (NIDA), and National Institute of
Mental Health (NIMH). The findings were reported
online in Nature on August 17, 2016.
Using computer modeling, the scientists evaluated
each compound in over 1 million structural
configurations to determine how well the molecules
can physically interact with the mu receptor. The
team chose 23 molecules for further pharmacological
testing.
The researchers then focused on optimizing the
structure of the most potent molecule, which
strongly activated the Gi pathway but had little
effect on beta-arrestin.
The resulting compound, called PZM21, was assessed
for its effects on pain in mice.
Mice treated with PZM21 showed pain relief
comparable to those treated with the opioid
morphine, but the effects lasted longer.
Unlike morphine, PZM21 did not slow the animals’
breathing, and it caused less constipation.
Further, PZM21-treated mice didn’t display the
drug-seeking behaviors of those given morphine,
suggesting that the drug may have less addictive
potential.
“This work demonstrates the power of structure-based
design to speed up the development of drugs with
optimal signaling and therapeutic properties”
explains Dr. Laurie Nadler, who heads NIMH’s
neuropharmacology program.
Further study will be needed to determine whether
PZM21 could serve as a safe and effective pain
reliever in people.
For more information
Structure-based discovery of opioid analgesics with
reduced side effects.
Manglik A, Lin H, Aryal DK, McCorvy JD, Dengler D,
Corder G, Levit A, Kling RC, Bernat V, Hübner H,
Huang XP, Sassano MF, Giguère PM, Löber S, Da Duan,
Scherrer G, Kobilka BK, Gmeiner P, Roth BL, Shoichet
BK.
Nature. 2016 Aug 17:1-6. doi: 10.1038/nature19112. [Epub
ahead of print]. PMID: 27533032.
Link...
MDN |